Residents and staff in care and nursing homes across the UK have been first in line to receive the new Covid-19 vaccinations. With the rollout of the vaccinations comes the hope that the end of lockdown restrictions are in sight and visitors will be able to return to see and hug their loved ones for the first time in many months.
We answer some of the questions that residents, staff and family members may have about the vaccinations in Country Court Care Homes.
The rollout of vaccinations into care and nursing homes has already started, with most residents and staff at Country Court Care and Nursing Homes having received the vaccination by mid-January. The priority of the vaccine rollout is determined by the JCVI (Joint Committee on Vaccination and Immunisation). The JCVI advises the UK government on immunisation. The vaccination programme is managed centrally by NHS England and locally by the CCG (Clinical Commissioning Group) that the care home falls under. The vaccination programme is administered by the GP surgery affiliated to the individual care home. Exactly when the care home residents receive their vaccination is dependent on how quickly a vaccine slot is allocated to the team from the GP surgery who will visit the care home. Typically, the GP surgery will try to give the care home manager 48-72 hours-notice, but this can vary.
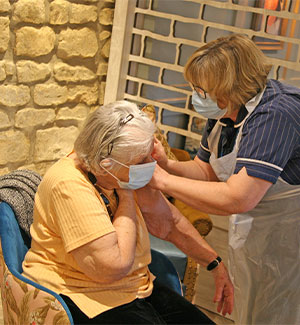
Before the COVID-19 vaccination is administered to residents in a care home a consent form needs to be signed by each person. This is either done by the resident themselves if they have the capacity or by a relative if they do not. The Manager of the care home will be able to tell you whether your relative has received the vaccination.
Care home workers are offered the vaccination either at their local vaccination centre e.g. at their local hospital hub or they may receive it at the same time as the residents in their care home. Arrangements differ locally according to which CCG the care home falls under.

The COVID-19 vaccination is not compulsory for care home staff but Country Court is proactively encouraging all staff to take up the offer of the vaccination. Country Court is delighted that the vaccine has been authorised and that residents and staff in their homes will be first in line to receive it.
The vaccines approved for use in the UK have met strict standards of safety, quality and effectiveness set out by the Medicines and Healthcare products Regulatory Agency (MHRA) which is an independent body. The coronavirus vaccination has been through all the clinical trials and safety checks that any other medication has to go through.

Most of the vaccines currently available are based on the fact that coronaviruses have club-shaped spikes on their outer coats, which form a corona – Latin for crown – on the virus surface. Studies suggest that the immune system is good at recognising these if it has encountered them before, and therefore these are a good approach for a vaccine. If a vaccine can produce a “spike” inside the body, the immune system recognises the “spike” and responds to it. That means that if the body encounters the Covid-19 virus in future, the immune system will recognise it and act against the virus so it cannot develop into an illness.
The Pfizer/BioNTech vaccine is an mRNA (messenger RNA) vaccine. Messenger RNA gives a body's cells instructions to make specific proteins - in this case, viral proteins that can be recognised by the immune system. This vaccine contains a small part of the genetic code for the SARS-CoV-2 spike protein. The vaccine delivers the mRNA to the body’s cells so that they make the protein that appears in the “spike” on the outside of the Covid-19 virus.
The vaccine does not contain any live virus and you cannot get coronavirus from having the vaccine.
The Oxford-AstraZeneca vaccine also contains the genetic sequence of this surface spike protein, so, like the Pfizer/BioNTech vaccine, when the vaccine enters cells inside the body, it uses this genetic code to produce the surface spike protein of the coronavirus. The difference is that instead of mRNA, the Oxford vaccine uses a harmless, weakened adenovirus (a different virus that also has the ‘spike’ protein on its surface). It has been genetically changed so that it can't cause an illness.
The Moderna vaccine works in a very similar way to the Pfizer/BioNTech vaccine. It uses mRNA to tell the body's cells to create the spikes that are on the surface of the coronavirus, so that the immune system can respond to them. It does not contain any live virus.
None of the approved vaccines contains any ingredients derived from animals. The Oxford-AstraZeneca vaccine uses harmless, weakened form of an adenovirus (a different virus that also has the ‘spike’ protein on its surface). The virus was grown in human embryonic stem cells. These stem cells weren’t taken directly from a human embryo – they are copies of stem cells which originally date from the 1970s. The vaccine itself does not contain any embryonic cells.
The vaccine is recommended by The British Islamic Medical Association and the Muslim Council of Britain. Imams have confirmed that all the vaccines are halal. The British Sikh community and Hindu leaders support the vaccine as do the Church of England and the Catholic Church, including the Oxford-AstraZeneca vaccine.
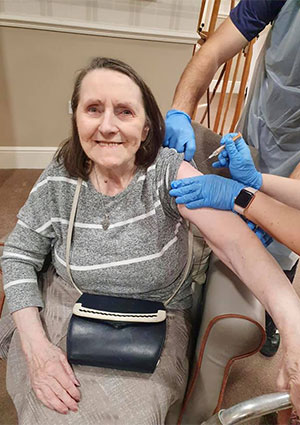
The version of the vaccine used depends upon local arrangements, but the majority of residents in Country Court Care Homes have received the Oxford-AstraZeneca vaccine as it is easier for local GP surgeries to store and transport. Where staff have received their vaccination at their local hospital hub this has often been the Pfizer/BioNTech vaccine.
The first does of the COVID-19 vaccine should give people good protection from coronavirus, the second dose is needed to give longer-lasting protection. It is too early to tell if people can get or spread coronavirus if they have had the vaccine, but indications are if this is the case it will be a weakened version. It is still important to adhere to social distancing guidelines and wear a mask where it’s hard to stay away from other people.
Most side effects of the COVID-19 vaccination are mild and should not last longer than a week, they may include a sore arm at the site of the injection, feeling tired, a headache, feeling achy or feeling or being sick. Anyone receiving the injection should tell the healthcare staff if they have ever had a serious allergic reaction (anaphylaxis).
Nurse Practitioners from the GP surgery that delivered the initial dose of the vaccine will revisit the care home 12 weeks after the delivery of the first dose to administer the second dose of the vaccine.
Care Homes must operate within the guidelines issued by their local Director of Public Health to find the right balance between the positive benefits of visits and the risk of transmission of COVID-19 to care staff and vulnerable residents. Country Court is very keen to allow visitors and families back into their care homes at the earliest opportunity. Families are a central part of life in our homes and have been sorely missed by both residents and staff. Visiting is crucially important for maintaining health and well-being and quality of life for residents and Country Court will allow visitors as soon as it is safe to do so.
Read more here: https://www.nhs.uk/conditions/coronavirus-covid-19/coronavirus-vaccination/coronavirus-vaccine/